Improve your clinical service, start using NeuroGuide’s reliable, fast & efficient Automatic Clinical Report Writer. No delays with minimal expense for a professional quality in-house QEEG report.
Automatic Clinical Reports
Price range: $350.00 through $800.00
Description
Automatic Clinical Reports for NeuroGuide’s ACR Writer
Improve your clinical service, start using NeuroGuide’s reliable, fast & efficient Automatic Clinical Report Writer today!
- No delays with minimal expense for a professional quality in-house QEEG clinical report
- Less than one minute to produce a professional QEEG clinical report, in Microsoft Word format, that a physician and/or clinician can edit
- ACR provides: Empowerment, simplicity, accuracy & efficiency!
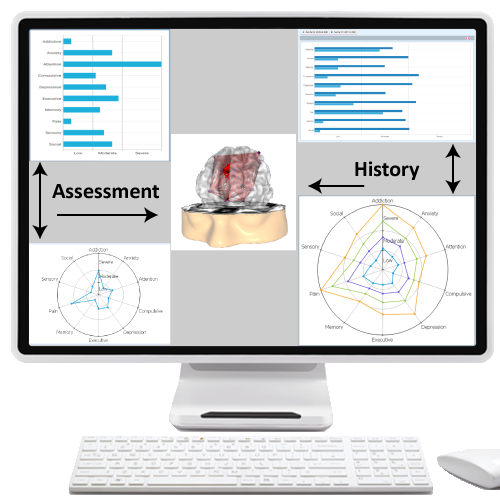
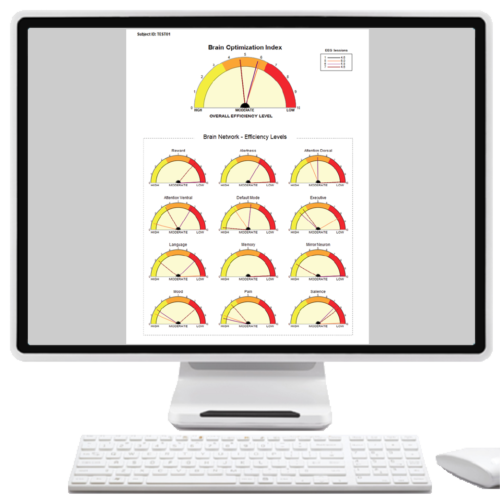
- Get valid Normative Database Comparisons using FDA compliant NeuroGuide™ software, without depending on internet Q-EEG report services!
- Get relevant content and displays, plus helpful NFB recommendations in less than a minute.
- Increased productivity by at least 10 Fold, e.g. ten reports in an hour!
This is How Easy it is to Use the ACR Writer:
- Select Artifact Free EEG & Verify High Test Re-Test Reliability
- Click Report > Automatic Clinical Report
- Enter your company name & contact information, add a logo if wanted and/or a scanned signature or select manual, click Save Information & then click OK. If you have already saved your Company Info, Logo & Signature, you can skip this step.
- Select Report Options, Desired Recommendations, Verify Artifact Free EEG and then click Generate Report
Click on the button below to Download the handy PDF that shows you:
- How to test to see if your computer is capable of generating ACRs;
- How to use the Demo Mode to Set up your Company information on the report to make sure the logo and signature is formatted correctly before using one of the ACRs you purchased to generate an actual reports.
Automatic Clinical Report writer QEEG reports are sold in packages. Choose 10 or 25 reports.
*Must Own NeuroGuide™ v 2.8.2 or greater to use ACR. Also, one must have the Learning Disabilities and Traumatic Brain Injury Discriminant Functions (DIS) (TBI & LD) and/or Brain Performance Index (BPI) add-on(s) on said NeuroGuide License if these features are to be included in the Report.
Additional information
| QEEG Reports | 10 QEEG Reports, 25 QEEG Reports |
|---|
Only logged in customers who have purchased this product may leave a review.




Reviews
There are no reviews yet.